Carbon dioxide (CO2) capture systems may be classified into three categories: post-combustion, pre-combustion, and oxy-combustion. The following figure illustrates those capture approaches, noting challenges, as well as established and developmental technologies in those areas.
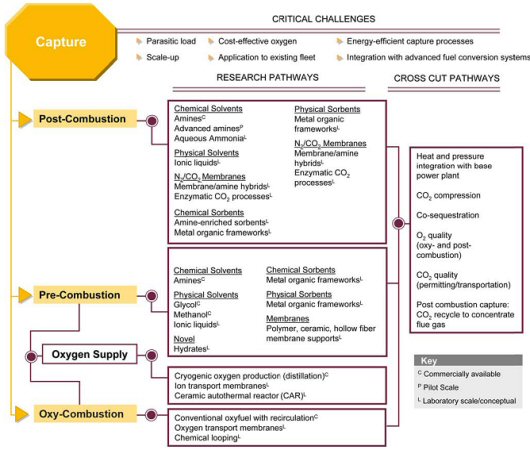
More detailed discussion on the post-combustion, pre-combustion, and oxy-combustion technologies for CO2 capture follows.
Post-Combustion CO2 Capture
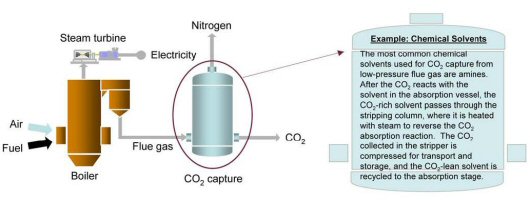
Post-combustion CO2 capture is primarily applicable to conventional natural gas and pulverized coal-fired (PC) power generation. In a typical PC power plant, fuel is burned with air in a boiler to produce steam, which drives a turbine to generate electricity. The boiler exhaust, or flue gas, consists mostly of nitrogen (N2), and CO2. Separating CO2 from this flue gas stream is challenging for several reasons:
- CO2 is present at dilute concentration (typically 13 to 15 volume percent for PC power plants and 3 to 4 percent for natural gas-fired plants) and at low pressure (slightly above atmospheric); thus, a large volume of gas has to be treated.
- Trace impurities (e.g., particulate matter, sulfur dioxide [SO2], nitrogen oxides [NO2] in the flue gas can degrade sorbents and reduce the effectiveness of certain CO2 capture processes.
- CO2 is captured at low pressure. Compressing it from atmospheric to pipeline pressure (about 2,000 psia) will incur a large auxiliary power load on the overall power plant system.
A post-combustion CO2 capture absorption process based on chemical solvents such as amines is pictorially shown below. These types of processes have been developed and deployed commercially in the refinery and chemical industries. To date, however, their use in PC power plants has been restricted to slipstream applications, and no definitive analysis exists as to the actual costs for a full-scale capture plant. However, conceptual baseline analyses prepared by NETL in 2022 assuming use of the Shell CANSOLV CO2 capture process (designed to recover high-purity CO2 from low pressure streams that contain O2, such as flue gas from coal-fired power plants, combustion turbine exhaust gas, and other waste gases) would raise the levelized cost of electricity from a new supercritical PC power plant by 66 percent, from 6.45 cents/kilowatt-hour (kWh) to 10.73 cents/kWh assuming 90% capture.
Pre-Combustion CO2 Capture for Gasification Application
Pre-combustion CO2 capture related to a gasification plant is pictorially depicted below. In gasification reactions, the amount of air or oxygen (O2) available inside the gasifier is carefully controlled so that only a portion of the fuel burns completely. This “partial oxidation” process provides the heat necessary to chemically decompose the fuel and produce synthesis gas (syngas), which is composed of hydrogen (H2), carbon monoxide (CO) and minor amounts of other gaseous constituents. The syngas is then processed in a water-gas-shift (WGS) reactor, which converts the CO to CO2 and increases the CO2 and H2 mole concentrations to about 40 percent and 55 percent, respectively, in the syngas stream.
At this point, the CO2 has a high partial pressure, which significantly improves the driving force for various types of separation and capture technologies. After CO2 removal, the H2 rich syngas is nearly pure hydrogen which can be deployed in multiple clean energy system applications. One application is to use H2 as a fuel in a combustion turbine to generate electricity in the context of an efficient combined cycle plant, where additional electricity is generated by extracting the energy from the combustion turbine flue gas via a heat recovery steam generator (HRSG). Other applications involve synthesis of liquid transportation fuels, ammonia or chemicals, while hydrogen itself can be deployed as a zero-carbon fuel.
In review, because the gasification process is operated at high pressure and CO2 is present at much higher concentrations in the syngas (i.e., in comparison with post-combustion flue gas), pre-combustion CO2 capture has the potential of being less expensive than post-combustion CO2 capture. For the same amount of CO2 captured, a much smaller volume of gas needs to be treated, leading to much smaller equipment size and lower capital costs.
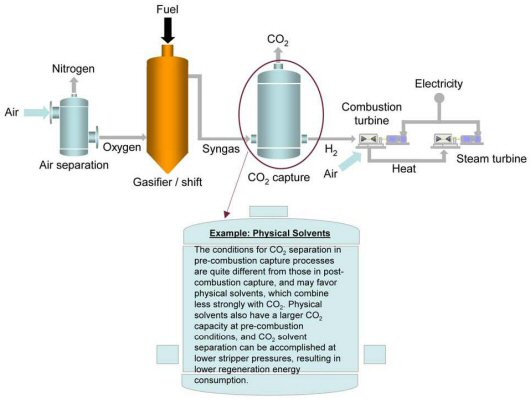
NETL has identified various research pathways for pre-combustion CO2 capture. Near-term applications of CO2 capture from pre-combustion systems will likely involve physical or chemical absorption processes, with the current state-of-the-art being a physical glycol-based solvent called Selexol. Mid-term to long-term opportunities to reduce capture costs through improved performance could come from membranes and sorbents currently at developmental stages. Recently updated (2022) baseline analysis conducted at NETL shows that CO2 capture and compression using Selexol raises the cost of electricity from a newly built GE quench gasifier-based IGCC power plant by about 37 percent, from 11.4 cents/kWh to 15.62 cents/kWh.
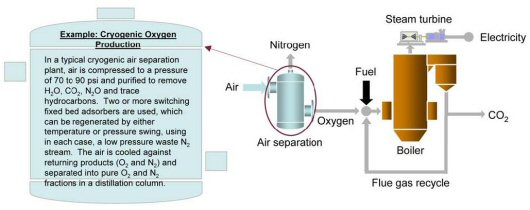
Oxy-Combustion CO2 Capture
The objective of pulverized coal oxygen-fired combustion is to combust coal in an enriched oxygen environment using pure oxygen diluted with recycled CO2 or water (H2O). The process is pictorially shown below. Under these conditions, the primary products of combustion are CO2 and H2O, and the CO2 can be captured by condensing the water in the exhaust stream. Oxy-combustion offers several additional benefits, as determined through large-scale laboratory testing and systems analysis:
- A 60-70 percent reduction in NOx emissions compared to air-fired combustion, mainly due to flue gas recycle, but also from reduced thermal NOx levels due to lower available nitrogen. Some nitrogen is still introduced into the system as nitrogen inherent to the coal matrix, and through air infiltration.
- Increased mercury removal. Boiler tests of oxy-fuel combustion using Powder River Basin (PRB) coal resulted in increased oxidation of mercury, facilitating downstream mercury removal in the electrostatic precipitator and flue gas desulfurization systems.
- Applicability to new and existing coal-fired power plants. The key process principles involved in oxy-combustion have been demonstrated commercially (including air separation and flue gas recycle).
Both pre-combustion and oxy-combustion utilize air separation to combust coal in an enriched oxygen environment. However, it is important to note that the amount of oxygen required in oxy-combustion is significantly greater than in pre-combustion applications, increasing CO2 capture costs. A higher purity oxygen stream would also need to be used. Oxygen is typically produced using low-temperature (cryogenic) air separation, but novel oxygen separation techniques involving innovative approaches that are cost effective at smaller scales are being developed to reduce costs.
Carbon Dioxide








