To date, the University of Southern Mississippi's synthetic research efforts have been focused on the development of stimuli-responsive water-soluble polymers designed for use in enhanced oil recovery applications. These model systems are structurally tailored for potential methods. The goal of previous synthetic work has been to design novel polymers that exhibit large dilute solution viscosities in the presence of the adverse conditions normally encountered in oil reservoirs (such as high salt concentrations, the presence of multivalent ions, and elevated temperatures).
Polyzwitterions were investigated in previous research. A goal of the current project is to investigate the interaction of surfactants with the hydrophobically modified (HM) polyzwitterions. Surfactants are critical components in micellar enhanced EOR processes because of their ability to reduce interfacial tension and mobilize oil trapped in reservoir formations. The aim is to synthesize polymer systems that will demonstrate synergistic increases in solution viscosity upon the addition of surfactants.
Project Results
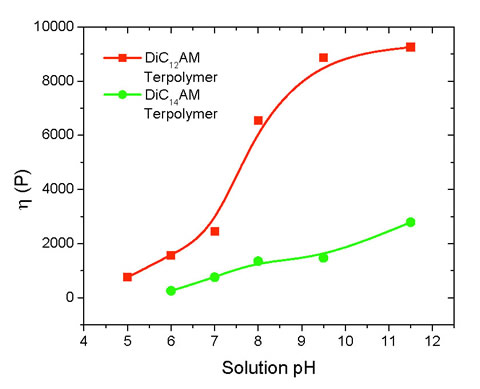
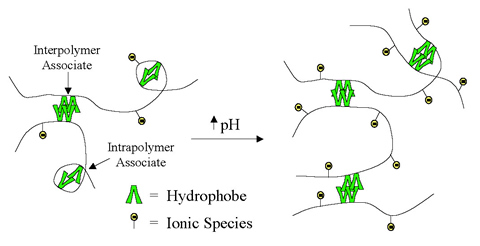
The new "smart" copolymers developed can control fluid mobility and/or conformance in oil recovery processes by altering viscosity and/or permeability using triggers based on pH, salt or temperature variations. The polymer systems developed may demonstrate superior performance as mobility control agents in micellar enhanced EOR processes due to surfactant-induced viscosity enhancement.
Benefits
The primary impact of "smart" copolymers will be to increase oil recovery by producing bypassed oil. The model developed has identified the parameters that are important to fluid mobility, thereby revealing methods to enhance solution performance when using polyions solutions as displacing fluids in oil reservoirs. The model was found to be valid for flexible polymer coils in aqueous salt solutions where intermolecular interactions are minimal. The "smart" polymers are designed to have "triggerable" properties such as changes in pH and/or salt concentration, which enable them to react to reservoir conditions to stimulate increased oil flow.
Project Summary
A new relationship based on Yamakawa-Fujii and OSF theories was developed to describe the contribution of electrostatic forces to the excluded volume of a polyelectrtolyte in solution. The model is valid for flexible polymer coils in aqueous salt solutions where intermolecular interactions are minimal.
As fluid passes through the channels of a porous medium, the fluid is continually accelerating and decelerating. A fluid flowing under these conditions experiences as extensional flow filed. The fluid drag forces that are applied to a polymer coil in a fluid under extensional flow are proportional to the average extension rate the fluid experiences. Although the fluid extension rate depends upon the porous medium's channel geometry it always increases, regardless of channel geometry, as the fluid flow rate increases.
When polymer solutions are used to flood an oil reservoir, very low fluid extension rates exist at large distances away from the injection well-head. To be effective in decreasing displacing fluid mobility during polymer flooding, polymer coils must experience extension as they percolate through the porous media. An understanding of which fluid flow conditions extend polymer coils is needed to control flooding and improve oil recovery.
In this study a hypothesis was formulated that presumes polymer coils will extend only when the rate of coil extension is greater than the rate of coil recovery. A mathematical analysis was used to develop a relationship that predicts the minimum fluid extension rate that produces coil extension. The minimum fluid extension rate was shown to be inversely proportional to the coils hydrodynamic diameter.
This finding implies that in typical reservoir flooding where fluid extension rates are very low, polymer coil extension that decreases displacing fluid mobility and improves oil recovery will occur only if the coil hydrodynamic diameter is extremely large. Large coil hydrodynamic diameters are formed when polymer molecular weights are high and the polymer solution intrinsic viscosity is large at the temperature conditions exist in the oil reservoir.